The Ore Itself
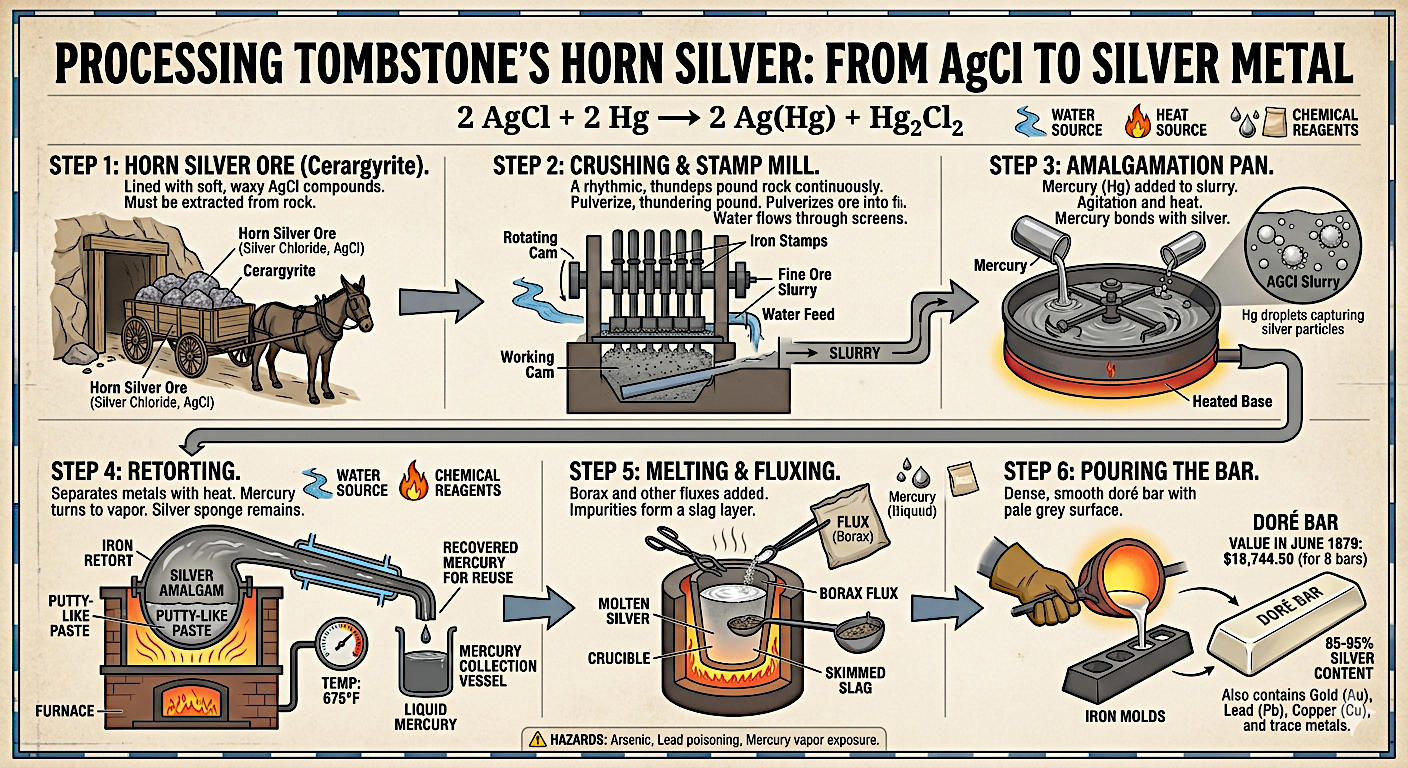
When Ed Schieffelin traced the Lucky Cuss vein in March 1878 and chipped off a sample, what he was holding was not pure silver. It was rock laced with silver compounds, primarily a mineral called cerargyrite, better known as horn silver. Horn silver is silver chloride (AgCl), a soft, waxy mineral that cuts like wax and turns a dark purplish-gray when exposed to sunlight. Miners recognized it immediately: it was heavy, it had a distinctive luster, and a good piece of it would leave a silver streak when scraped across a surface.
The upper ore zone at Tombstone was dominated by these oxidized silver minerals, formed over millions of years as rainwater and oxygen worked down through the rock and chemically altered the original silver sulfide deposits. The resulting silver chloride ores were exceptionally rich, but they were still locked inside rock. Liberating the silver required several stages of work, most of it done not at the mines themselves but at the mills strung along the San Pedro River nine miles away, in the towns of Charleston and Millville.
Getting the Ore to the Mill
Once blasted loose underground, ore was hoisted to the surface in iron buckets or ore cars and sorted by grade at the surface. High-grade ore went directly to the mill. Lower-grade material might be stockpiled or discarded as waste. Sorting was skilled work: an experienced miner could look at a piece of rock and judge its silver content within a reasonable range simply by its weight, color, and texture.
From the mines south of town, the ore traveled nine miles by wagon to the mills. Teams of mules or horses hauled the ore in heavy freight wagons over rough roads, a journey that was slow, dusty in summer, and muddy in the rainy season. The mills were built on the San Pedro River rather than at the mines because milling required large quantities of water for the stamp mills and amalgamation pans, and the river provided it reliably. The mills at Charleston and Millville processed many millions of dollars worth of Tombstone ore during the boom years. The man who had confirmed the bonanza through assay at the Brunckow fireplace also built the first of those mills. Gird traveled to San Francisco to supervise construction of the original quartz mill at Millville, had it erected at the Fulton Iron Works, and returned to run it as superintendent — the same technical capacity, now applied at every stage from ore sample to finished bar.
That arrangement began to shift in July 1881, when the mines struck water at roughly 520 feet in the Sulphuret shaft. As pumps were installed to fight the flooding, large quantities of water came to the surface with every hour of operation. Entrepreneurs recognized that this pumped water could power mills right at the mine head, eliminating the expensive nine-mile freight haul entirely. The Girard Mill, built in Tombstone with twenty stamps, did exactly that. By using mine water rather than paying to haul ore to the San Pedro, it saved approximately $3.50 per ton compared to the Charleston mills, a meaningful margin when processing hundreds of tons of rock each week. The water problem that threatened to shut the mines down also, briefly, made milling cheaper.
By late 1883 the mill had become part of Tombstone's industrial life. The diarist George Parsons recorded visiting it that October with a group of friends: "Went through the Grand Central Mine this A.M. with Arthur, John and Sam, and later through Girard Mill." The pairing of the underground mine tour with a visit to the surface mill captures how fully integrated the two operations were in the minds of long-time Tombstone residents.
The Stamp Mill
The first step at the mill was crushing. Stamp mills were the workhorse of nineteenth-century ore reduction, and their sound was the background noise of every silver and gold camp in the American West: a rhythmic, thundering pound that could be heard for miles.
A stamp mill worked on a simple principle. Heavy iron stamps, each weighing several hundred pounds, were lifted by a rotating cam and dropped onto the ore below, over and over, sixty to one hundred times per minute. The ore was fed into the crushing chamber continuously, and water flowed through to wash the pulverized material forward through a screen. Only particles fine enough to pass through the screen moved on to the next stage; anything too coarse was crushed again. The result was a wet slurry of very fine rock powder, gray and heavy, carrying the silver compounds suspended in it.
The Tombstone district mills ran anywhere from ten to twenty stamps each. The sound of a twenty-stamp mill running at full capacity was extraordinary: a sustained, deafening roar that rattled windows and could be heard in the hills above town.
Amalgamation: Mercury Does the Work
Once the ore was crushed to a fine slurry, the challenge was separating the silver from the rock powder. For silver chloride ores, the solution was mercury. Mercury has a remarkable property: it readily combines with silver and gold to form an alloy called amalgam, while leaving the surrounding rock powder alone. A miller could pour mercury into a pan of ore slurry, agitate the mixture, and the mercury would seek out and bond with the silver particles, pulling them out of suspension.
The amalgamation process at the Tombstone mills typically used large iron pans, heated from below, in which the ore slurry and mercury were mixed and stirred by mechanical arms. The heat and agitation helped the mercury contact every silver particle in the slurry. After sufficient mixing time, the heavier amalgam settled to the bottom of the pan while the depleted rock slurry, called tailings, was drained off. The amalgam was then collected.
The chemistry happening in the pan was straightforward. Mercury reacted with the silver chloride to produce silver amalgam and mercurous chloride, a white powder also known as calomel:
2 AgCl + 2 Hg → 2 Ag(Hg) + Hg2Cl2
The silver was now captured in the amalgam. The trick was getting it back out.
Retorting: Driving Off the Mercury
Amalgam at room temperature looks like a dull gray putty or paste, soft and heavy. It contains roughly equal parts silver and mercury by weight, plus trace amounts of whatever else the mercury picked up. To recover the silver, millers had to separate the two metals, and the method was heat.
The amalgam was loaded into a sealed iron vessel called a retort, which was then placed in a furnace and heated to around 675 degrees Fahrenheit. At that temperature, mercury boils and turns to vapor, while silver, with a much higher boiling point, stays behind as a solid. The mercury vapor traveled through a pipe into a water-cooled condenser, where it returned to liquid and dripped into a collection vessel to be reused. Mercury was expensive and toxic; mills were careful to recover as much of it as possible.
What remained in the retort after the mercury was driven off was a porous, sponge-like mass of impure silver. Millers called it retort silver or silver sponge. It was not attractive, and it was not yet a finished product. The silver was there, but it was riddled with air pockets and still contained small amounts of lead, copper, and other metals that had come along for the ride.
The Final Step: Pouring the Bar
The silver sponge was transferred to a crucible and placed in a high-temperature melting furnace. Borax and other fluxes were added to help draw off impurities. As the silver melted, the fluxes combined with the base metal contaminants and floated to the surface as a layer of slag, which was skimmed away. What remained in the crucible was molten silver, still not quite pure but close enough for commercial purposes.
The molten silver was poured into iron molds and allowed to cool. The result was a doré bar: a dense, smooth, slightly rounded bar of silver with a pale gray surface. "Doré" is a French word meaning gilded or golden, and the name reflects the fact that these bars were not pure silver. They typically contained 85 to 95 percent silver, with the remainder being gold (always present in small amounts in Tombstone ore), lead, copper, and other trace metals. The exact composition varied with the ore body and the efficiency of the mill's operation.
The first doré bars from the Tombstone district were driven to Tucson on June 17, 1879 by Ed Schieffelin and Judge Bidwell in the same blue spring wagon used to arrive at the mines. There were eight bars, valued at $18,744.50. They were displayed at the Safford, Hudson and Company bank in Tucson, where they drew considerable attention from a town that had been skeptical of Schieffelin's claims from the beginning.
What Happened to the Bars
Doré bars were an intermediate product. Before the silver could be used for coinage or industrial purposes it had to be refined further to remove the remaining impurities. The bars were shipped to smelters and assay offices, eventually making their way to the United States Mint. There, the silver was refined to near-purity, assayed for exact content, and paid for at the prevailing market rate, less the cost of refining and shipping.
The gap between the mine and the mint was where fortunes were made or lost. A mine that produced ore assaying at $15,000 per ton, as the Lucky Cuss did, was extraordinarily rich. But even ordinary ore at $100 or $200 per ton could be profitable if the mill was efficient and the silver price held steady. When the price of silver began its long decline through the 1880s, the math that had made Tombstone's largest operations so valuable gradually stopped working. The San Pedro mills were the first to feel it: the Charleston mill was dismantled in May 1886. In 1887, fires destroyed the Grand Central and Contention pumping works, flooding those two great mines permanently. But other mines, including the Tough Nut, continued producing into the twentieth century, and from 1901 to 1911 the Tombstone Consolidated Mines Company made a serious attempt to drain and reopen the flooded lower levels. The milling process described here remained essentially unchanged throughout that entire period. What changed was the economics: no amount of milling efficiency could compensate for silver prices that had fallen by half since the boom years. For more on why the mines ultimately closed, see Why Did The Mines Close?
Hazards of the Mill
The mills at Charleston and Millville were productive, but they were not safe. Workers at the furnaces faced serious occupational hazards every day. Richard Fulton, writing in the Journal of Arizona History in 1966 based on firsthand accounts from mill workers, described the furnace as "a deadly place to work." Furnacemen were continuously exposed to fumes from arsenic and lead, both present in the ore alongside silver. Workers who developed poisoning had few options: the folk remedy was to curl into a specific body position with pillows pressed against the stomach. The only physician believed capable of treating the condition practiced in Park City, Utah, hundreds of miles away. Some men avoided the furnace entirely because of its reputation.
Mercury presented a separate and less visible danger. During retorting, mercury vapor filled the air around the furnace before being drawn into the condenser. Mills made every effort to recover the mercury, both for its high cost and because experienced millers understood its toxicity, but complete capture was impossible. Prolonged exposure to mercury vapor causes damage to the nervous system, kidneys, and lungs, a condition known today as acrodynia or, in its chronic form, as Minamata disease. Workers in 19th-century amalgamation mills across the American West showed recognizable symptoms without any medical framework to explain what was happening to them.
The environmental cost fell on the San Pedro River. Fulton describes the river valley before the mills as a lush riparian corridor with catfish and other fish, willows and cottonwoods, and abundant wildlife. Mill tailings, discharged into or near the river after amalgamation, carried residual mercury, lead, and arsenic into the water and sediment. Mercury contamination from 19th-century amalgamation mills has been documented throughout the American West and remains detectable in riverbeds more than a century after the mills closed. The San Pedro below Charleston would have received the same legacy.
Sources
- Richard W. Fulton, "Millville-Charleston, Cochise County 1878–1889," Journal of Arizona History, vol. 7, no. 1 (Spring 1966), pp. 9–22. Source for furnace worker health hazards and the pre-mill condition of the San Pedro River valley.
- Levi F. Butler, David D. Wilson, and Dix Rasor, "Geology and Ore Deposits of the Tombstone District, Arizona," Arizona Bureau of Mines Bulletin No. 143 (1938). Source for ore mineralogy, production figures, and mill operations.
- Herbert Love, History of Tombstone to 1887 (1933). Source for mill locations, workforce details, and the first silver shipment.
- Lonnie E. Underhill, ed., "The Tombstone Discovery: The Recollections of Ed Schieffelin and Richard Gird," Arizona and the West, vol. 21, no. 1 (Spring 1979), pp. 37–76. Source for the first silver bars, Gird's role in building the Millville mill, and assay values at discovery.
- George W. Parsons, Journal, Volume 2 (June 28, 1882 – March 31, 1887), Arizona Historical Society. Source for the October 1883 visit to the Grand Central Mine and Girard Mill.
